[ad_1]
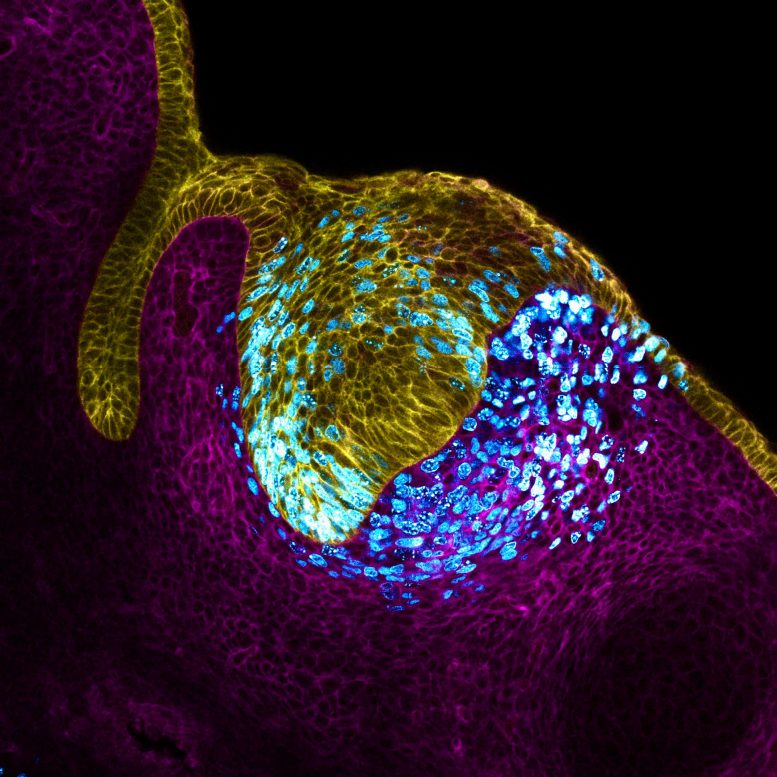
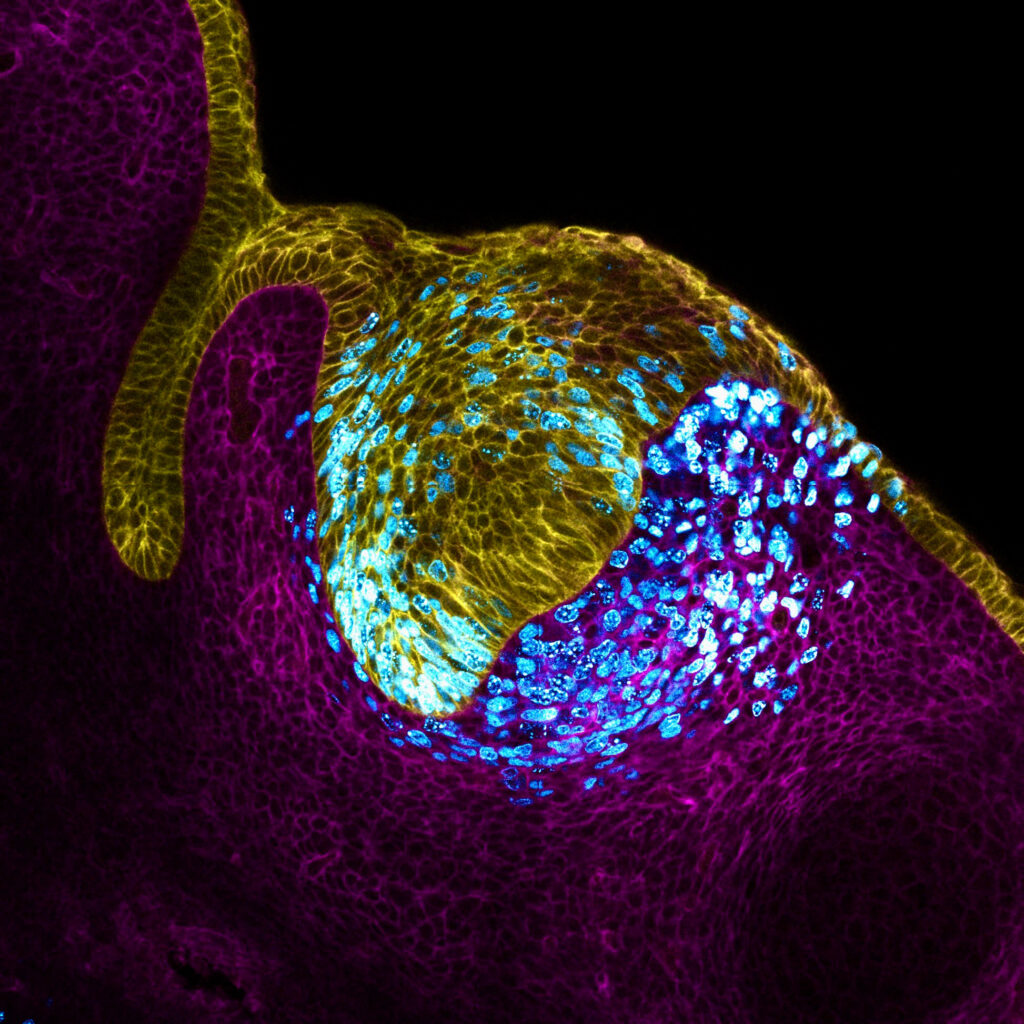
Tooth epithelium (cell surface; yellow) and stroma (cell surface; magenta). Proliferating cells (cyan) expand the tissue, creating mechanical pressure at the center of the tissue, driving the formation of the primary tooth signaling center or organizer (enamel knot).Image source: Neha Pincha Shroff and Xu Pengfei
Without a map, finding your way around the winding streets of some cities can be a real challenge. To determine our directions, we rely on a variety of information, including digital maps on our phones and identifiable shops and landmarks. Cells in our bodies face similar problems when building organs during embryogenesis. They need instructions on where to go and how to behave. Fortunately, like cell phone towers in a city, embryos have specialized cells in specific locations, called organizers, that send signals to other cells and help them organize themselves to build our complex organs.
Some of these signals are sent by elements from the organizer (a privileged signaling center). Cells around it receive stronger or weaker signals depending on its location and make decisions accordingly. The wrong location of these information centers in tissues can lead to fatal embryonic malformations. Scientists have long known how these signaling centers are related, but how they appear in specific locations has remained elusive.
Discovered through international collaboration
It took an international collaboration of physicists and biologists to find the answer. A few years ago, the laboratories of Professor Ophir Klein at Cedars-Sinai Guerin Children’s and the University of California, San Francisco (UCSF) and Professor Otger Campàs at the TU Dresden and the Cluster of Excellence in Physics of Life at the University of California, Santa Barbara UCSB figured out how it worked and banded together. Together, they discovered that mechanical pressure within the growing tissue determines where the signaling centers emerge.
“Our work shows that both mechanical stress and molecular signaling play a role in organ development,” said Ophir Klein, MD, executive director of Cedars-Sinai Guerin Children’s, the David and Meredith Kaplan Distinguished Chair in Children’s Health and the Co-corresponding author of the study.
Mechanical stress in tissue cells
The study was published in Nature Cell Biology, Research shows that as cells grow in embryonic incisors, they sense increasing pressure and use this information to organize themselves. “It’s like one of those toys that grows in size when it absorbs water,” said Dr. Neha Pincha Shroff, a postdoctoral fellow at the UCSF School of Dentistry and co-first author of the study. “Imagine this happening in a confined space. What happens in the incisor node is that cells multiply in a fixed space, which causes pressure in the center, and then a specialized group of cells forms. Like people in a crowded bar. Likewise, cells in the tissue began to feel the squeeze from their peers, and the researchers found that the cells that felt the stronger pressure stopped growing and began sending signals to organize other cells around the tooth. They were literally forced to become tooth organizers. .
“We were able to use microdroplet technology previously developed in our laboratory to figure out how the accumulation of mechanical stress affects organ formation,” said Otger Campàs, Ph.D., professor and co-corresponding author of the study and now managing director. Chair of Excellence in Tissue Dynamics at the Physics of Life Cluster and former associate professor of mechanical engineering at the University of California, Santa Barbara. “It’s really exciting that tissue stress plays an important role in establishing signaling centers. It will be interesting to see if or how mechanical stress affects other important developmental processes.
The embryo uses several of these signaling centers to direct cells to form tissues and organs. Like building a skyscraper or a bridge, shaping our organs requires rigorous planning, a lot of coordination, and correct structural mechanics. Failure in any of these processes can be catastrophic when building a bridge, and it can also cause damage to us as we grow in the womb.
“By understanding how embryos form organs, we can begin to ask what goes wrong in children with congenital malformations,” said Ophir Klein. “This work may lead to further research into how birth defects develop and are prevented.”
Reference: “Proliferation-driven mechanical compression induces signaling center formation during mammalian organ development” Authors: Neha Pincha Shroff, Pengfei Xu, Sangwoo Kim, Elijah R. Shelton, Ben J. Gross, Yucen Liu, Carlos O. Gomez , Qianlin Ye, Tingsheng Yu Drennon, Jimmy K. Hu, Jeremy BA Green, Otger Campàs and Ophir D. Klein, April 3, nature cell biology.
DOI: 10.1038/s41556-024-01380-4
The research was funded by the National Institute of Dental and Craniofacial Research (OK and OC), the German Research Association under the German Excellence Strategy, and the Biophysics Excellence Cluster (OC) of the Technical University of Dresden.
[ad_2]
Source link